

This workbook walks students through calculating enthalpies for the A Level energetics topic in a structured, active way — gap-fills, scaffolded calculations, and exam-style questions that build confidence without handing them the answers. It’s designed to keep students thinking and writing throughout the lesson, not just passively reading slides.
Topics covered:
- Enthalpy changes — exothermic and endothermic reactions, enthalpy profile diagrams
- Standard conditions and standard enthalpy definitions (combustion, formation, neutralisation, reaction)
- Calorimetry — determining enthalpy of combustion experimentally using q = mcΔT
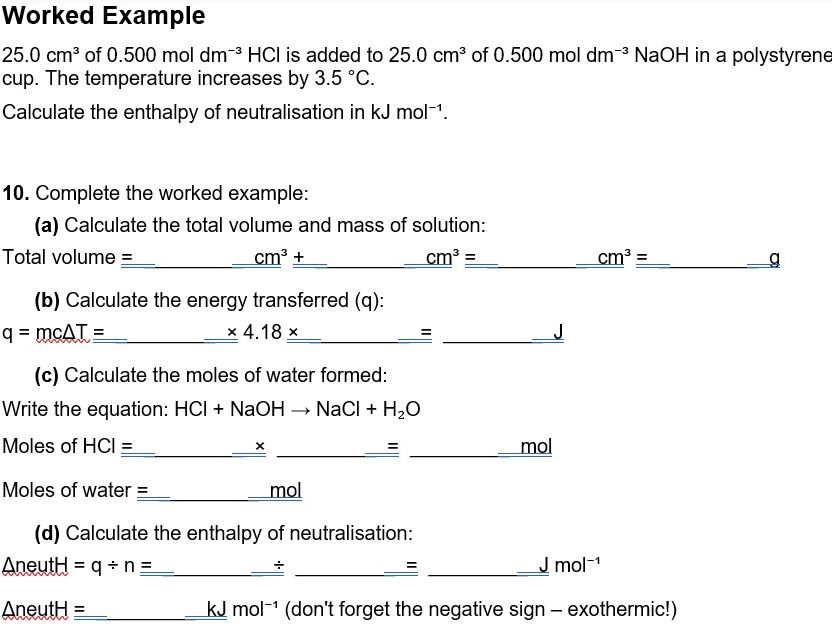
- Enthalpy of neutralisation — coffee cup calorimetry method with graphical analysis
- Bond enthalpies — calculating enthalpy changes, average bond enthalpies, limitations of the method
- Working backwards to find unknown bond enthalpies from experimental data
Key features:
- Gap-fill activities throughout, reinforcing key definitions and concepts without overwhelming weaker students
- Step-by-step worked examples with guided calculation spaces (mass → moles → q → ΔH)
- Exam tips embedded at point of use — e.g. enthalpy profile diagram conventions, structural formula reminders
- Limitations and improvements tables for practical work (a common exam question students often fumble)
- Full section of exam-style questions (3–6 markers) mapped to OCR A (H432) command words
- Self-assessment checklist at the end for student reflection
- Aligned to OCR Chemistry A Module 3.1.1 (Enthalpy Changes) and 3.1.2 (Bond Enthalpies)
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have downloaded this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.
£0.00