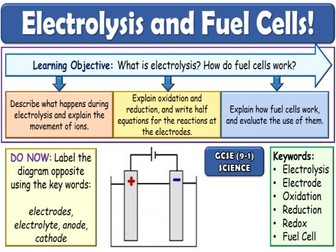
Rates of Reaction GCSE Chemistry
Perfect if you teach GCSE Chemistry Single Science, this fully differentiated and resourced lesson supports your students in learning about rates of reaction. Covering the full GCSE Chemistry specification, this resource is adapted to cover all exam boards. Students will:
State what is meant by rate of reaction.
Calculate the rate of a reaction.
Describe the different experimental methods used to investigate the rate of a reaction - disappearing cross experiment, change in mass experiment, volume of gas given off experiment.
Explain what is meant by collision theory and state what factors the rate of a reaction depends on.
Explain how changes in temperature, concentration, pressure and surface area affect the rate of reaction.
Complete rate experiments involving gases and precipitates.
Describe what a catalyst is and explain how it affects the rate of a reaction.
This lesson includes a detailed and engaging lesson PowerPoint with differentiated activities, class practical/demo activities, exam style questions and progress quizzes for students to complete. Enjoy and feel free to leave a review.
*INCLUDED IN BUNDLE:
24 slide PowerPoint Presentation with key points, progress check questions, checkpoints, practice exam questions, quizzes, class practical activities. *