
This booklet takes one of the most demanding topics at A Level — Born-Haber cycles and lattice enthalpy — and scaffolds every definition, every calculation, and every conceptual link (ionic radius, ionic charge, hydration enthalpy) so students build understanding step by step rather than drowning in simultaneous new ideas. With gap-fills that force active engagement while the worked examples model exactly what examiners expect to see.
Topics Covered:
- Lattice enthalpy of formation — definition, always exothermic, as a measure of ionic bond strength
- Lattice enthalpy of dissociation — the reverse process, same magnitude but positive sign
- Comparing lattice enthalpies — NaCl vs NaF, NaCl vs MgO, using ionic radius and charge to explain differences
- Key enthalpy definitions needed for Born-Haber cycles: enthalpy of formation, atomisation, 1st and 2nd ionisation energy, 1st and 2nd electron affinity
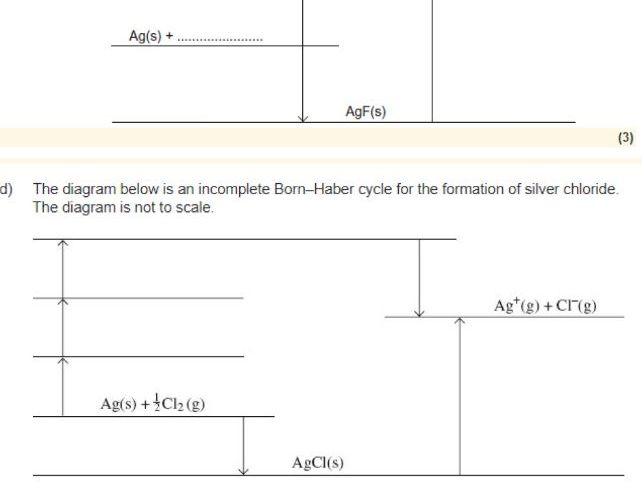
- Born-Haber cycle structure — how all the enthalpy changes connect, the key formula (ΔfH = ΔatH + IE + EA + ΔLEH)
- Worked example: calculating lattice enthalpy of CaO (including 2nd IE and 2nd EA)
- Worked example: calculating enthalpy of formation of MgBr₂ (with the “watch out — two bromine atoms” trap)
- Enthalpy of solution — definition, the two-step model (break the lattice, then hydrate the ions)
- Enthalpy of hydration — definition, always exothermic, relationship to ion–water attractions
- The dissolution enthalpy cycle: ΔsolH = –ΔLEH(formation) + ΣΔhydH
- Worked example: enthalpy of solution of MgCl₂
- Effect of ionic charge and ionic radius on lattice enthalpy and hydration enthalpy — qualitative explanations
Key Features
- Gap-fill definitions for every key enthalpy term — students aren’t just reading definitions, they’re constructing them, which is far better for retention
- 2nd electron affinity explicitly taught and explained (adding an electron to an already negative ion requires energy to overcome repulsion) — a concept students routinely get wrong in exams
- Lattice enthalpy data table with six compounds of increasing complexity (NaCl → Al₂O₃) — students analyse trends in ionic charge and radius before doing any calculations
- Worked examples with partially completed calculations — specific values left blank so students fill in the numbers at each algebraic step, modelling the working examiners want
- The MgBr₂ example deliberately flags the stoichiometric trap of doubling atomisation and electron affinity values for two bromine atoms — catches the exact mistake students make in exams
- Full answers section at the back — includes gap-fill answers and all numerical solutions with working, so students can self-mark independently
- Designed for UFP Chemistry students but equally usable for OCR A / AQA A Level Chemistry.
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have downloaded this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.