
This booklet tackles one of the trickiest required practicals at GCSE — titration — by breaking the method, the reasoning, and the salt preparation into distinct sections that build logically on each other. It starts by establishing why titration is even necessary (you can’t just add excess alkali and filter), which gives students a reason to care about the technique before they learn the steps.
Topics covered:
- Starter retrieval on general acid reactions — acid + metal, acid + base, acid + metal carbonate word equations
- Sulfuric acid reactions with magnesium, magnesium oxide, magnesium hydroxide, and magnesium carbonate
- Why titration is needed — the problem of soluble alkalis that can’t be filtered out, requiring an exact volume instead
- The titration method step by step — pipette, burette, indicator, adding acid drop by drop, reading the end-point
- The ionic equation for neutralisation — H⁺(aq) + OH⁻(aq) → H₂O(l), plus spectator ions
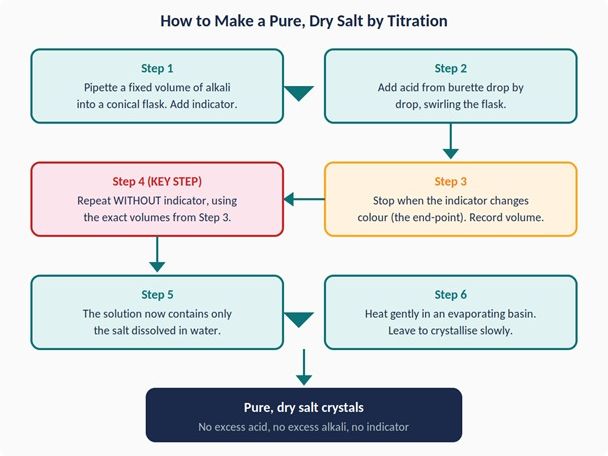
- Making a pure, dry salt — repeating the titration without indicator, evaporation, and crystallisation
- Why universal indicator is unsuitable — gradual colour change makes the end-point impossible to identify precisely
Key features:
- Opens with a clear conceptual rationale for why titration exists — contrasts it with the “add excess and filter” method used for insoluble reactants, so students understand the logic before touching the equipment
- Word banks provided for every gap-fill section, keeping the booklet accessible for EAL learners and lower-ability students
- Labelled diagram of the titration setup paired with the step-by-step method — students can connect the written instructions to the physical apparatus
- Sequencing activity where students order the salt preparation steps 1–6 — tests procedural understanding in a format that mirrors how examiners ask it
- Tip boxes embedded throughout explaining key points (e.g. why the second titration is done without indicator, why phenolphthalein or methyl orange are preferred over universal indicator)
- Sentence starters provided for longer exam-style answers — models how to open an explanation without giving the answer away
- Stretch and challenge question on why titration is better than direct reaction for making sodium chloride and potassium chloride — pushes higher-ability students to think about reactivity
- Exam-style questions ranging from 1-mark equipment recall up to multi-mark explanations of the full salt preparation procedure
- Self-assessment checklist with three confidence levels (Confident / Mostly / Not yet) tied to eight specific skills, plus a reflection prompt
- Aligned to Edexcel Combined Science / Chemistry GCSE specification CC8e
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have downloaded this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.