
This booklet breaks Hess’s Law down into a clear, repeatable method — combustion cycles and formation cycles taught separately, with partially completed calculations that force students to engage with each algebraic step rather than just copying a model answer. It’s particularly strong on the “why” behind Hess’s Law, starting with the problem (some enthalpy changes can’t be measured directly) before introducing the solution, which gives the topic purpose from the outset.
Topics covered:
- Why Hess’s Law is needed — reactions that don’t go to completion or produce multiple products
- Standard enthalpy of formation (ΔfH⦵) — definition, writing balanced equations for exactly one mole of product
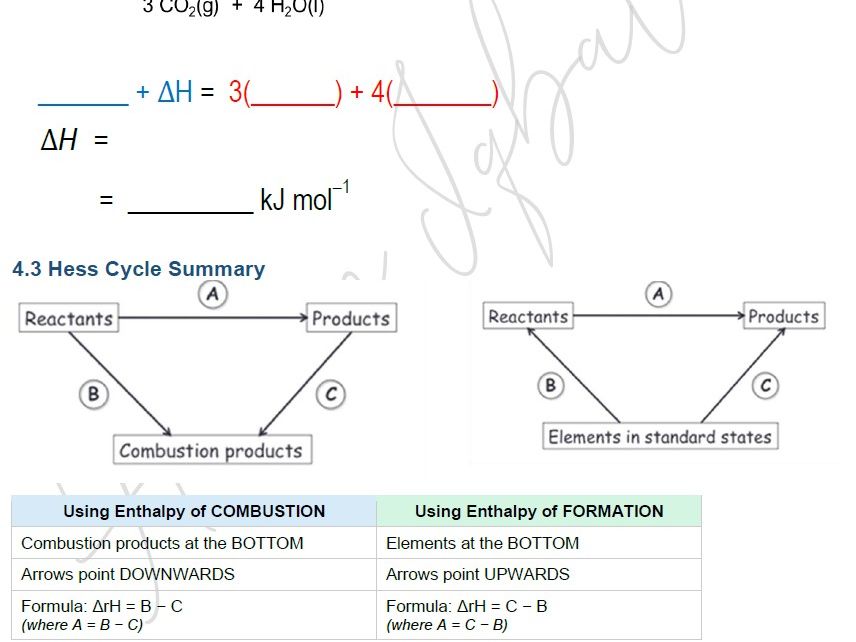
- Hess cycles using enthalpy of combustion data — combustion products at the bottom, arrows pointing downwards
- Hess cycles using enthalpy of formation data — elements at the bottom, arrows pointing upwards
- Worked examples: formation of ethanol (combustion data), combustion of propane, combustion of methane (formation data), oxidation of SO₂
- Unfamiliar Hess cycles — constructing cycles from given intermediate equations (e.g. N₂ → NO → NO₂)
- Enthalpy of neutralisation — calculating ΔneutH from experimental data including limiting reagent identification
- Coffee cup calorimetry — procedure, graphical method for temperature change, limitations vs combustion experiments
Key features:
- Specification-mapped to OCR Chemistry A (H432) Module 3.2.1, with the spec reference printed on the front page so students know exactly where this fits
- Key vocabulary box upfront — defines Hess’s Law, enthalpy cycle, ΔfH⦵, ΔcH⦵, and standard conditions before any calculations begin
- Summary comparison table for combustion vs formation cycles — what goes at the bottom, which direction the arrows point, and the formula for each — a revision-ready reference students can return to
- Partially completed calculations in worked examples — students fill in specific values and algebraic steps rather than starting from a blank page
- Exam tips embedded at point of use (e.g. ΔfH⦵ of elements = 0, equations must show formation of exactly one mole, fractional coefficients are allowed)
- Practice builds from guided gap-fills through to fully independent Hess cycle construction (draw it yourself, calculate it yourself)
- Exam-style questions in Part C covering all key question types: combustion cycles, formation cycles, unfamiliar intermediate data, neutralisation calculations, and a 6-mark extended response on experimental procedure
- Self-assessment checklist tied to six specific learning objectives rather than broad topic headings
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have downloaded this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.