
This booklet walks students through the full titration practical — from preparing a standard solution to calculating the relative formula mass of an unknown organic acid — with every step written out explicitly and the calculation broken into manageable chunks. It’s built to work as a live lesson companion, not just revision material, with EAL tip boxes that explain the language of practical chemistry (what “persists” means, why we rinse, how to read a meniscus) without dumbing down the science.
Topics covered:
- Indicator selection — comparing methyl orange and phenolphthalein (pH ranges, colour changes), choosing the right one for a strong acid–strong alkali titration
- Preparing a standard solution — weighing by difference, dissolving, transferring to a volumetric flask, rinsing, and making up to the mark
- The titration method — rinsing pipette and burette with the correct solutions, performing a rough titration, then accurate titrations until concordant results are obtained
- Recording results — burette readings to the nearest 0.05 cm³, results table with rough, titre 1–3, and mean titre
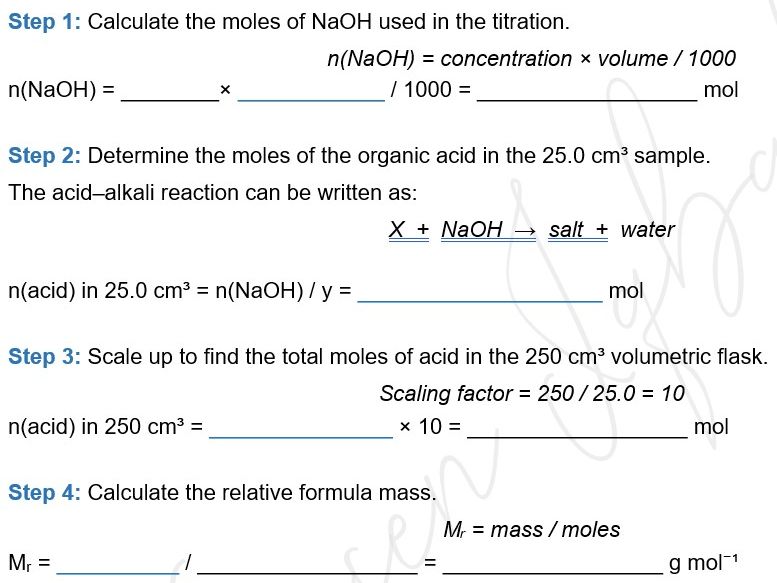
- Calculating Mr from titration data — moles of NaOH → moles of acid (using stoichiometry) → scaling up from 25.0 cm³ to 250 cm³ → Mr = mass/moles
- Identifying an unknown organic acid — matching the calculated Mr against succinic, fumaric, malic, and tartaric acid
Key features:
- Full equipment list at the top — students can check they have everything before starting, which avoids disruption mid-practical
- Safety precautions box covering goggles, lab coats, and NaOH handling — ticks the risk assessment box without taking up half the booklet
- EAL tip boxes at every critical point: what a standard solution actually is, why we rinse the beaker into the volumetric flask, how to read a burette at eye level, what “persists” means in context — these explain the practical vocabulary that native speakers take for granted
- Step-by-step calculation scaffold with blanks at each stage — students fill in the values rather than staring at a blank page, which models the working examiners want to see
- Distinction between rough and accurate titrations made explicit — including the technique of adding quickly until 2 cm³ before the rough titre, then switching to drop-by-drop
- Concordant titre guidance built in — only averaging values within 0.10 cm³, with a reminder not to include the rough
- Table of organic acids (succinic, fumaric, malic, tartaric) provided for identification — turns the calculation into a genuine problem-solving task where students match their Mr to the correct compound
- Gap-fill exercise reinforcing key terminology (standard solution, meniscus, titre, end point, concordant)
- Exam-style questions targeting common practical assessment points: white tile reasoning, rinsing rationale, not blowing out the pipette, improving mass accuracy
- Aligned to OCR Chemistry A (H432) practical skills and Module 3.2.1
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have downloaded this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.