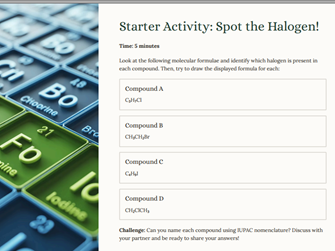

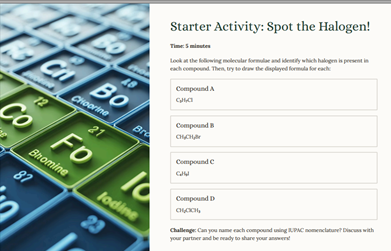
**This Powerpoint specifically uses the Cambridge AS Level Chemistry Syllabus (9701) and covers all criteria for – Unit Halogenoalkanes
**
Students will be able to -
1 recall the reactions (reagents and conditions) by which halogenoalkanes can be produced:
(a) the free-radical substitution of alkanes by Cl2 or Br2 in the presence of ultraviolet light, as exemplified
by the reactions of ethane
(b) electrophilic addition of an alkene with a halogen, X2, or hydrogen halide, HX(g), at room temperature
© substitution of an alcohol, e.g. by reaction with HX(g); or with KCl and concentrated H2SO4 or
concentrated H
3PO4; or with PCl 3 and heat; or with PCl 5; or with SOCl 2
2 classify halogenoalkanes into primary, secondary and tertiary
3 describe the following nucleophilic substitution reactions:
(a) the reaction with NaOH(aq) and heat to produce an alcohol
(b) the reaction with KCN in ethanol and heat to produce a nitrile
© the reaction with NH3 in ethanol heated under pressure to produce an amine
(d) the reaction with aqueous silver nitrate in ethanol as a method of identifying the halogen present as exemplified by bromoethane
4 describe the elimination reaction with NaOH in ethanol and heat to produce an alkene as exemplified by bromoethane
5 describe the S
N1 and SN2 mechanisms of nucleophilic substitution in halogenoalkanes including the inductive effects of alkyl groups
6 recall that primary halogenoalkanes tend to react via the SN2 mechanism; tertiary halogenoalkanes via the S
N1 mechanism; and secondary halogenoalkanes by a mixture of the two, depending on structure
7 describe and explain the different reactivities of halogenoalkanes (with particular reference to the relative strengths of the C–X bonds as exemplified by the reactions of halogenoalkanes with aqueous silver nitrates)
**Every Powerpoint includes learning objectives, a starter and plenary, suitable activities and review questions with mark scheme. We also provide a free pdf version of the file so it can easily be printed for students as a handout or uploaded to the school VLE system
**
Bring your Cambridge AS Level Chemistry lessons to life with our expertly designed teaching resources. Each unit is tailored to the latest syllabus, offering ready-to-use lesson plans, engaging experiments, and visually clear notes that save hours of prep time. Whether you’re new to the course or refining your approach, The Science Shelf gives you everything you need to boost student understanding and exam success — all in one place.
Get this resource as part of a bundle and save up to 50%
A bundle is a package of resources grouped together to teach a particular topic, or a series of lessons, in one place.
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have purchased this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.