
Directly aligned to the OCR Chemistry A H432 specification with every key syllabus point clearly covered.
Fully editable document.
Written in clear, accessible language while including all essential technical keywords students must use in exams.
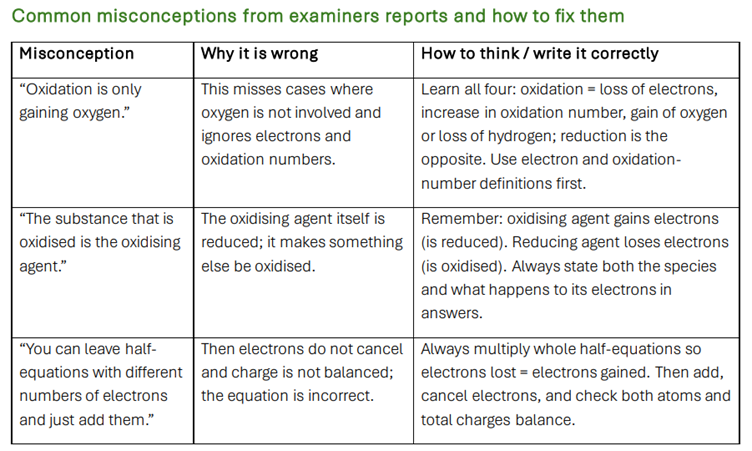
Strong emphasis on common misconceptions taken from examiner reports, with clear corrections and model wording to prevent mark loss.
Designed to work for focused, specification-led revision.
Specification points covered:
• Definitions of oxidation and reduction using electrons, oxygen, hydrogen and oxidation numbers
• Oxidising and reducing agents explained accurately in terms of electron transfer
• Rules for assigning oxidation numbers, including common exceptions
• Constructing and balancing redox equations using half equations in acid and alkali
• Redox titrations: Fe²⁺/MnO₄⁻ and I₂/S₂O₃²⁻ systems, including mole ratios and calculation structure
• Standard electrode potentials, standard conditions and the standard hydrogen electrode
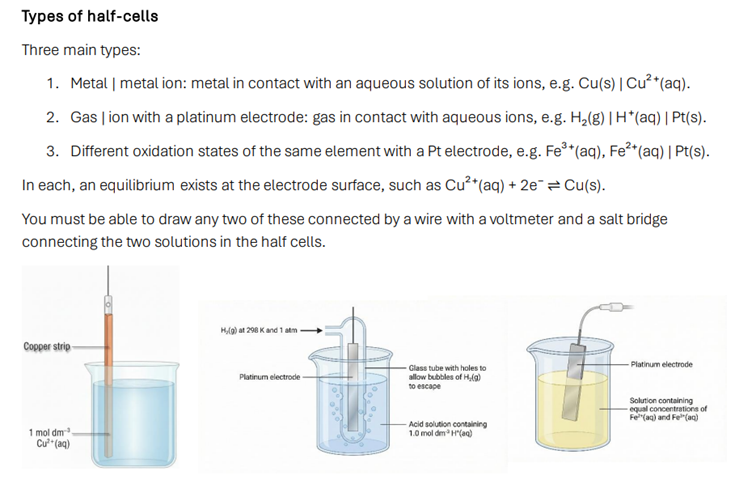
• Writing and interpreting half cells and full electrochemical cells
• Calculating E°cell correctly using electrode data
• Using E° values to predict feasibility and understanding limitations of predictions
• Practical set up of cells including salt bridge and voltmeter
• Storage cells, rechargeable cells and fuel cells
• Hydrogen fuel cell reactions, advantages, disadvantages and evaluation points
Comprehensive revision resource for A Level Chemistry students!
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have purchased this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.