

How do we write reaction schemes? How are Grignard reagents used to increase the length of a carbon chain? What is the mechanism for Grignard reactions? All this and more covered in this comprehensive lesson with questions and answers! This is a Year 13 A level lesson for Edexcel International Unit 5 – WCH15, but it can also be used for all UK exam boards. All the slides in this lesson are fully animated and include answers to every mini plenary question and exam question. The breakdown of the slides (which are best opened on Microsoft PowerPoint) is as follows:
Slide 1 - Title and 5-minute starter. The starter is a grid of four questions entitled ‘last week, last lesson, today’s learning and future learning’. Use this generic slide for all of your lessons by simply changing the questions and the answers each time.
Slide 2 - Lesson objectives (see thumbnail image)
Slide 3 – Hinge question: What is synthesis?
Slide 4 – Worked Example 1: How can bromoethane be converted to ethyl propanoate? The solution to this problem appears on the screen as you click.
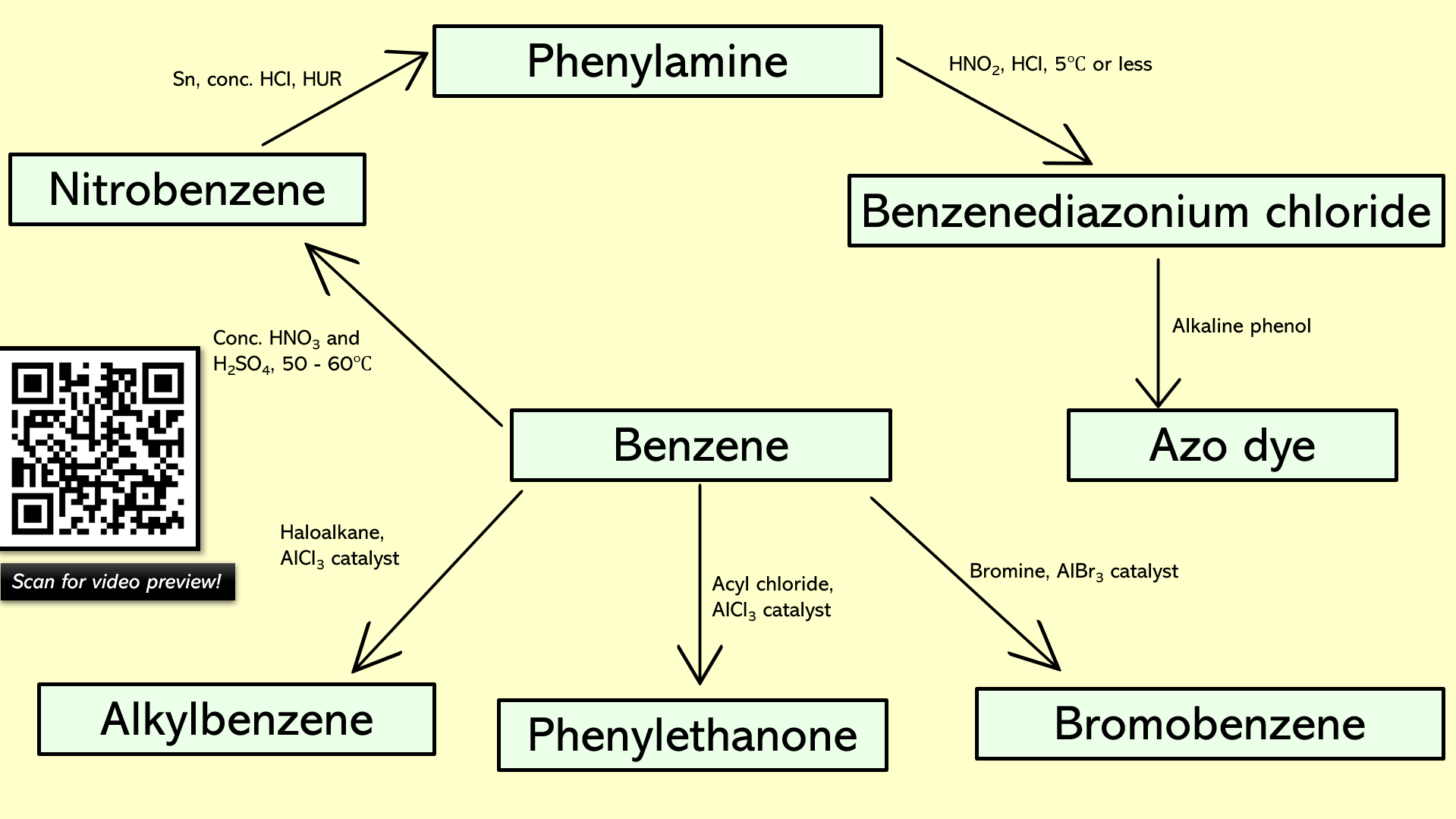
Slides 5 – 23: Planning for an organic synthesis. There are nine conversion diagrams for students to complete. Print for the students and help them write the conditions/reagents on each arrow. All the answers appear as you click
Slide 24 – Exploring the four ways to extend a carbon chain
Slides 25 – 29: Grignard reagents are presented to students, covering all the following: what they are, how they are formed, a mechanism for the formation of a Grignard reagent and its reaction with methanal, conditions for Grignard reactions and a list of the four different Grignard reactions (with carbon dioxide, methanal, aldehyde of two carbons or more and a ketone)
Slides 30 – 33: Two worked examples for Grignard reactions with full working out as you click
Slide 34 – Mini plenary. Two questions to check understanding of Grignard reactions.
Slides 35 – 36: Predicting the chemical properties of unfamiliar compounds with familiar functional groups, using an example of phenylmethanol and 4-methylphenol
Slides 37 – 41: a 4-step reaction synthesis, very challenging problem! Full working out appears as you click
Slides 42 – 48: Exam questions with mark scheme answers (included with the resource)
If you have a positive experience with the resource, please leave a positive review! This really helps promote our store! Thanks!
Something went wrong, please try again later.
Really clean, professional layout that isn't cluttered with unnecessary text. The diagrams for the functional group conversions are some of the best I've seen—much clearer than the ones in our current textbook. Saved me a ton of formatting time. Thank you!
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.