
States of Matter: Solid, Liquid, and Gas
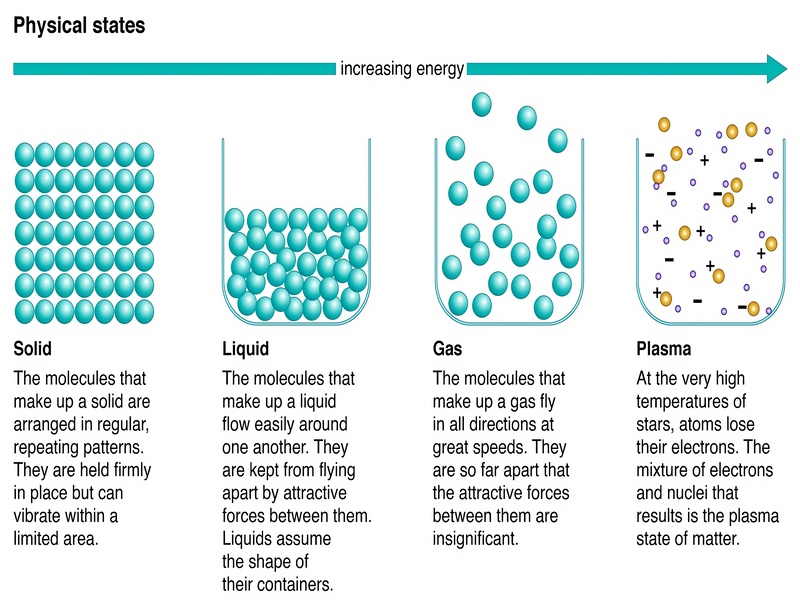
Everything around us, from the air we breathe to the chair we sit on, is made of matter. Matter can exist in different forms, which scientists call “states of matter” or “phases of matter”. You probably already know about the three main states of matter that we see every day: solids, liquids, and gases. But there’s also a fourth common state called plasma, which you might have seen in neon signs or even lightning.
What makes these states different? It all comes down to how the tiny particles (like atoms or molecules) that make up the matter are arranged and how they move. Think of it like a dance party: in a solid, everyone is packed tightly together, barely moving from their spots. In a liquid, people are still close but can move around and slide past each other. In a gas, everyone is spread out and moving freely and quickly.
Let’s look closer at solids. In a solid, the particles are held very close together in fixed positions. This is why solids have a definite shape and a definite volume. Imagine a block of ice; it keeps its shape and takes up the same amount of space no matter where you put it. The particles in a solid mostly just vibrate in place.
Liquids are a bit different. Their particles are still close together, but they can move past one another. This allows liquids to flow and take the shape of their container, though they keep a fixed volume. Think about pouring water into a glass; the water fills the bottom of the glass and takes its shape. Liquids and solids are sometimes called “condensed phases” because their particles are so close together.
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have purchased this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.