John Baum's Shop
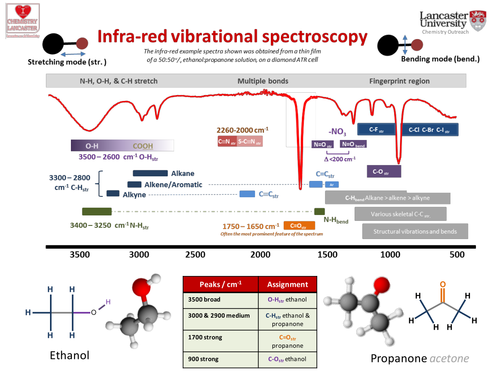
Exercises and resources in Chemistry. These resources have been used in various outreach activities for Chemistry / STEM master classes and visit days. Any confusion or comments let me know I believe you can message on this system or via the website. I am particularly interested in developing resources for Chemistry Out reach (AS /A2) , STEM activities, and competitions for lower school (yr 9+), so any suggestions or offers of collaboration gratefully received.