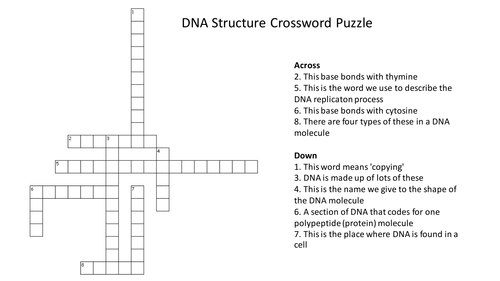
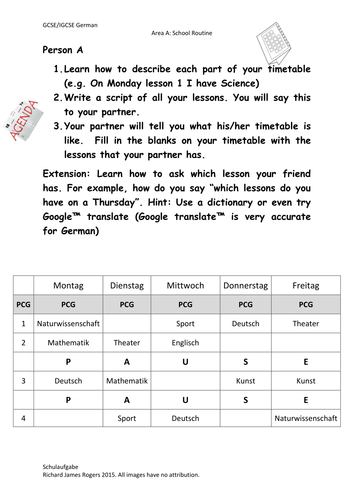
Richard Rogers' Shop
Author of 'The Quick Guide to Classroom Management: 45 Secrets That All High School Teachers Need to Know' which is available at https://www.amazon.com/dp/1798536722/ Currently teaching IBDP and IGCSE Chemistry at an international school in Bangkok, Thailand. I'm originally from North Wales in the UK.