


This short revision lesson reminds students how to use the atomic and mass numbers to identify numbers of protons, electrons and neutrons. The lesson should only take about 10-15 minutes and has been primarily designed for use with those students taking the AQA GCSE Combined Science FT in the lead up to mock and GCSE examinations, but is also suitable for students taking the HT, or separate chemistry.
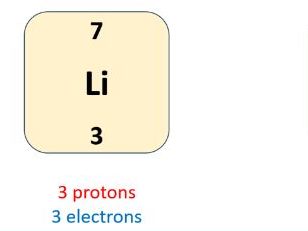
The lesson begins with a reminder that the atomic number represents the number of protons in a substance, and if that substance is an atom, the number of protons and electrons will be equal. Moving forwards, the students are reminded how to calculate the number of neutrons and then a series of examples are modelled to ensure that any misconceptions or mistakes are addressed. The students are provided with some understanding checks in the style of exam questions and all answers are embedded into the PowerPoint to allow students to assess their understanding and progress. Calculation of the numbers of subatomic particles is challenged on the GCSE examination papers most years, so this is a good opportunity for students to pick up marks. The key points associated with the topic have also been displayed on a resource which can be kept with the understanding checks for use in the final days and hours before the exam.
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have purchased this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.