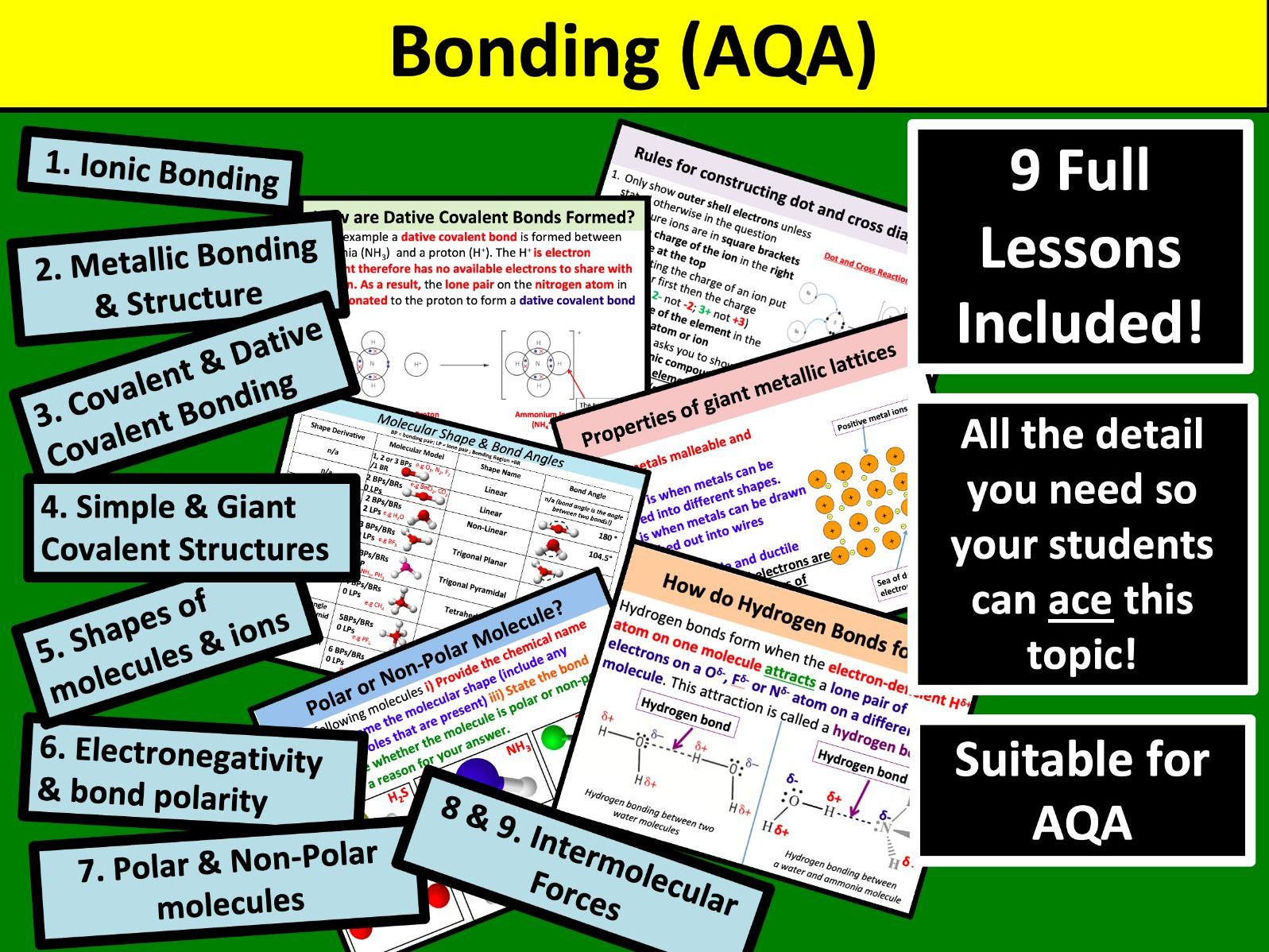
Resources included (9)

Electronegativity & Bond Polarity

AS Chemistry: Ionic Bonding

AS Chemistry: Intermolecular Forces (Part 1)

Shapes of Molecules and Ions (AQA)

Polar and Non-Polar Molecules

Metallic Bonding and Structure

Simple & Giant Covalent Structures

AS Chemistry: Intermolecular Forces (Part 2)

Covalent and Dative Covalent Bonding
A 9 full lesson bundle covering the AQA AS Level Chemistry Chapter: Bonding. Suitable for the AQA Specification.
If you teach OCR or Edexcel please purchase this bundle instead: https://www.tes.com/teaching-resource/resource-12425984
Lesson 1: Ionic Bonding
- To know ionic bonding as electrostatic attraction between positive and negative ions, and the construction of ‘dot-and-cross’ diagrams
- To explain solid structures of giant ionic lattices are a result of oppositely charged ions strongly attracted to each other in all directions
- To link the structure and bonding of ionic compounds on their physical properties including melting and boiling points, solubility and electrical conductivity in solid, liquid and aqueous states
Lesson 2: Metallic Bonding and Structure
- To describe the structure of metals
- To explain metallic bonding as strong electrostatic attraction between cations and delocalised electrons
- To explain the physical properties of giant metallic structures
Lesson 3: Covalent and Dative Covalent Bonding
- To know covalent bonding as electrostatic attraction between a shared pair of electrons and the nucleus
- To construct dot and cross diagrams of molecules and ions to describe single and multiple covalent bonding
- To apply the term average bond enthalpy as a measurement of covalent bond strength
- To know what a dative covalent bond is
- To construct dot and cross diagrams of molecules and ions to describe dative covalent bonding
Lesson 4: Simple and Giant Covalent Structures
- To describe the structure of simple and giant covalent compounds
- To explain how the structure and bonding of simple and giant covalent compounds link to their different physical properties
- To evaluate the potential applications of covalent structures based on their physical properties (stretch & challenge)
Lesson 5: Shapes of Molecules and Ions
- To determine the number of bonding pairs & lone pairs in a molecule or ion
- To recall the shapes and bond angles of molecules and ions with up to six electron pairs surrounding the central atom
- To explain the shapes of molecules and ions using the electron pair repulsion theory
- To construct diagrams to illustrate the 3D shapes of molecules and ions
Lesson 6: Electronegativity and Bond Polarity
- To define the term electronegativity
- To explain the trend in electronegativity down a group and across a period
- To explain what a polar covalent bond is bond and to illustrate this type of bond in a molecule
Lesson 7: Polar and Non-Polar Molecules
- To describe the difference between polar and non-polar molecules
- To explain why non-polar molecules can contain polar bonds
- To predict whether molecules are polar or non-polar
Lesson 8 : Intermolecular Forces (Part 1)
- To understand intermolecular forces based on induced-dipole interactions and permanent dipole-dipole interactions
- To explain how intermolecular forces are linked to physical properties such as boiling and melting points
- To compare the solubility of polar and non-polar molecules in polar and non-polar solvents
Lesson 9 : Intermolecular Forces (Part 2)
- To understand hydrogen bonding as intermolecular forces between molecules containing N, O or F and the H atom of –NH, -OH or HF
- To construct diagrams which illustrate hydrogen bonding
- To explain the anomalous properties of H2O resulting from hydrogen bonding
Declaimer: Please refrain from purchasing this popular resource for an interview lesson or a formal observation. This is because planning your own lessons including using your own lesson PowerPoints is a fundamental skill of a qualified/unqualified teacher that will be reviewed during these scenarios outlined above
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have purchased this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.