
C9-Organic chemistry-Hydrocarbons lesson created in accordance to the NEW AQA Specification (9-1). Designed for a high ability year 11 separates class, although content can be adjusted to suit any ability. Includes: slide animations, embedded videos and practice questions with answers on slides as well as a interactive review task. If for any reason the video link does not work, a URL has also been included in the notes.
AQA spec link: 4.7.1.1
Crude oil is a finite resource found in rocks. Crude oil is the remains of an ancient biomass consisting mainly of plankton that was buried in mud.
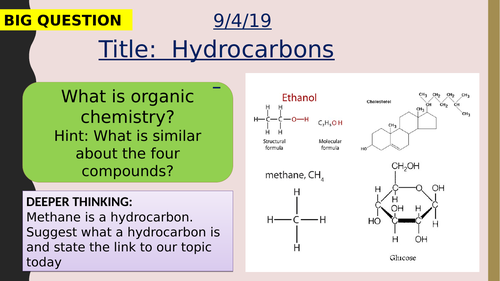
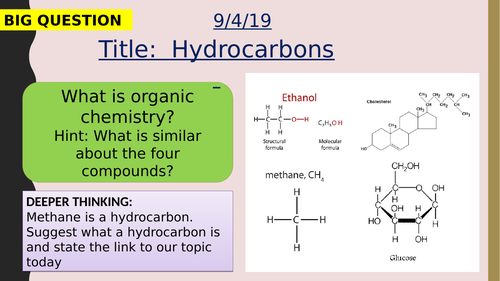
Crude oil is a mixture of a very large number of compounds. Most of the compounds in crude oil are hydrocarbons, which are molecules made up of hydrogen and carbon atoms only.
Most of the hydrocarbons in crude oil are hydrocarbons called alkanes. The general formula for the homologous series of alkanes is CnH2n+2
The first four members of the alkanes are methane, ethane, propane and butane.
Alkane molecules can be represented in the following forms: C2H6 or Students should be able to recognise substances as alkanes given their formulae in these forms.
Students do not need to know the names of specific alkanes other than methane, ethane, propane and butane.
Get this resource as part of a bundle and save up to 22%
A bundle is a package of resources grouped together to teach a particular topic, or a series of lessons, in one place.
Something went wrong, please try again later.
This resource hasn't been reviewed yet
To ensure quality for our reviews, only customers who have purchased this resource can review it
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.