















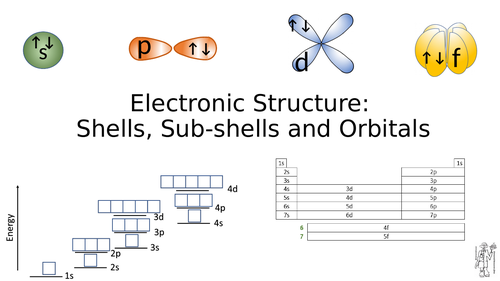


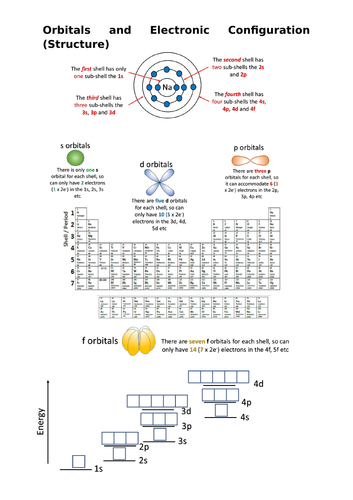
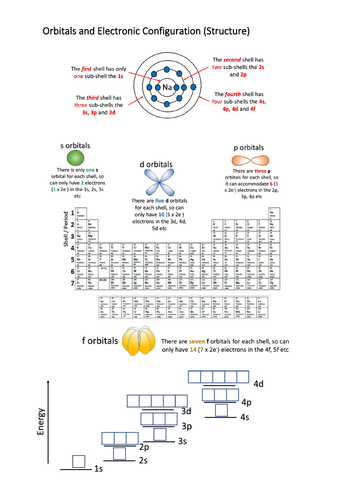



This resource explains how to write the electronic configuration of the first 36 elements in terms of their shells, sub shell and orbitals. It covers the AQA syllabus, but can be used with others. It contains a 30+ page Power Point which explains how electrons fill up the lowest energy levels first, how electrons prefer to stay unpaired and the rather tricky transition metal 4s/3d energy levels.
It explains how to write the configuration for neutral atoms as well as ions and contains a page of questions, based around exam questions from AQA. Questions have comments from the examiners report to say how difficult students in the exam found this type of question.
It also contains:
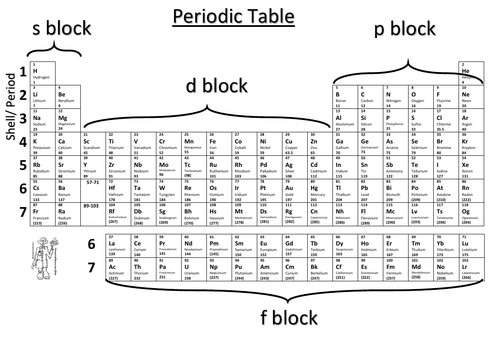
Periodic Table with all configurations
Student sheet with diagrams on for students to add notes to (or cut out and stick in diagrams)
Help sheet to practice configuration
Question sheet with answers provided on the Power Point
Homework/Extra Questions sheet to challenge the most able
An animated slide that shows all 36 configurations on an energy level diagram and position on the Periodic table.
Other AS lessons are available:
Ionisation energy:https://www.tes.com/teaching-resource/ionisation-energy-as-level-12412339
TOF Spectrometer and Isotope Calcuations: https://www.tes.com/teaching-resource/mass-spectrometer-tof-and-isotope-calculations-12386980
Oxidation States: https://www.tes.com/teaching-resource/oxidation-states-lesson-as-level-12421177
Rules for Electron Configuration Sheet can be found here for free:
https://www.tes.com/teaching-resource/electronic-configuration-rules-for-s-p-d-a-level-12406254
Please feel free to visit my shop where there are free and paid resources on science at:
https://www.tes.com/teaching-resources/shop/stephenkemp193
Please comment if you have found this resource useful.
Thank you
Something went wrong, please try again later.
Excellent resource - very clear and matches AQA specification
Excellent resource with good scaffolding techniques
Report this resourceto let us know if it violates our terms and conditions.
Our customer service team will review your report and will be in touch.